Restricting the spread of
insects, vehicles for virus transmission (≡
vector
control) is unavoidable in preventing and localizing
infections. As for
the way of managing it, let's have a look at the experiences
collected
by other continents.
1. In their
publication at the turn of this century, researchers
from Cuba and Venezuela [Rodriguez Coto M.M. et
al.(2000): Malathion Resistance in Aedes aegypty
and
Culex quinquefasciatus After its Use in
Aedes aegypty Control Programs J. Amer. Mosquito Control
Association, 16: 324-330.] reported on the stable
resistance to chlorinated hydrocarbons (like the fat soluble,
carcinogenic, antiandrogenic and teratogenic dichlorodiphenyltrichloroethane-DDT,
with characteristics of
increasing ion permeability across membranes) in two species
of
insects; in Aedes aegypti and in Culex quinquefasciatus.
After DDT, the next period of insecticides favoured the use
of neurotoxic organic phosphates (OPs)
targeting enzyme
acethylcholinesterase. Among OPs it
is temephos (acting on insect larva ≡ larvicide),
phenitrothion and malathion (acting on adult insect ≡
adulticide)
that had widespread use in practice. It was of great surprise
that
in spite of the intense and robust spraying of
OPs in the Caribbean area in the preceding 15
years, the resistance to malathion of the Aedes aegypti populations
remained at a low or slightly moderate level. Therefore, in restraining
Dengue fever the vector control further relied on the use of malathion. Opposite to Aedes aegypti, populations of Culex quinquefasciatus developed
strong resistance to malathion. This resistance is explained
by the upregulation of insect genes coding for detoxifying esterases, more precisely, by
the appearance of two esterase isoenzymes in Cuba, Venezuela, Columbia,
Brazil, with different migration patterns in acrylamide gel
electrophoresis.
Resistance probably introduced by migration or passive dispersal was
neither confirmed nor excluded. No data were found of the resistance
developed to insecticides similar to natural pyrethrins,
the Pyrethroids acting on voltage-gated Na+ channels in nerve
cell
axon membrane.
2. A Brazilian-French research
is reported in a publication of 2007 [da
Costa-Ribeiro M.C.V. et al.(2007): Low Gene Flow of
Aedes aegypti between Dengue-Endemic and Dengue-Free Areas in
Southeastern and Southern Brazil Am. J. Trop. Med.
Hyg. 77: 303–309.], comparing the southeast
(Dengue fever is
endemic) region to the southern (Dengue fever free) region in Brazil.
For
the population genetic follow up of passive gene transfer, populations
of Aedes aegypti
were collected in 11 and 1 towns of the regions
respectively.
Samplings were performed in highly populated localities
minimum
1,2 km and
maximum 1,538.6 km in distance from one another, and, they all have
ground
transport connections to Rio de
Janeiro. The sampling period lapsed from March 2003 - December 2003, in
three-month intervals, with regard to rainy and dry seasons. The insect
samples were cared for under standard circumstances (temperature,
humidity, parts of day...) until reaching the adult stage in
development.
For biomarkers in the samples, isoenzyme (> genetic)
polymorphism
was chosen
covering the same 10 enzymes per each of the samples (among them
glucose-phosphate
dehydrogenases, hexokinases, glycerophosphate dehydrogenases...). For
isoenzyme polymorphism the reference strain Paea/Tahiti (French
Polynesia/1994) was used
in the electrophoretic pattern analysis. The evaluation of degree
in differentiation, the indication of genetic variability in
populations, was made according to the Hardy-Weinberg principle.
It was determined, that Aedes aegypti populations
under study were highly differentiated
with changing patterns according to the rainy and the
dry
seasons, further, depending on transitions in weather
periods and more, on habitat. Since genetic variability was found also
in samples collected nearby the ground transport connections
(autoroutes), the conclusion was drawn that regions of Brazil
under study could not give rise to passive transmission of
Dengue. Transmission of Dengue is performed by insect
populations sensitive for the virus, that is they become
carriers,
however, according to theories, there is no opportunity for
virus
carrier state in insect strains of high and changeable
genetic variability [Gooding
R.H.(1996): Genetic Variation in Arthropod
Vectors of Disease-Causing
Organisms: Obstacles and Opportunities Clinical
Microbiology Reviews 9: 301–320.].
3. A report on the resistance
status of Aedes albopictus collected in some
areas of the United States was published in 2014 [Marcombe S. et
al.(2014): Insecticide Resistance Status of United
States Populations of Aedes albopictus and Mechanisms Involved
PLoS ONE 9(7): e101992.
doi:10.1371/journal.pone.0101992]. Insect populations collected
on the East Coast (five
counties in New Jersey, one county in Pennsylvania, two counties in
Florida) were studied in F1 progeny populations
(eggs-larvae-pupae-adults) for the effect of 11 insecticidal compounds
(6
of them larvicids and 5
of them adulticids) by monitoring two main mechanisms of developing
resistance; target mutations and the increased expression of
detoxifying enzymes. Results were referred to those observed
in an insect
population (ATM95) sensitive to all the chemicals applied. It was
demonstrated
that larval populations of all the eight sampling sources had
no
strong resistance to the insecticides studied. Temephos (group OP of
insecticides) proved to be effective in every larval populations of all
sampling sources. Nevertheless, worth to consider the strong resistance
to malathion (group OP of insecticides) in adult populations
from Florida and, adult populations
from New Jersey were already on the way to reach this kind of resistance.
All
the insect populations were sensitive to Pyrethroids.
Concurrently, DDT
resistance could be demonstrated in samples from Florida. This latter
suggests the feasibility of developing cross resistance to Pyrethroids
too,
in Florida region, since dispersal of Aedes albopictus in the USA
was preceded by the ban of DDT use (1972), further, targets for
Pyrethroid action are in overlap with targets for DDT action
(> voltage-gated ion channels). So, resistance to DDT
of Aedes albopictus throwed shadow on the
promising effects of Pyrethroids. Anyway, resistance to DDT in
Florida region could be a consequence of passive transfer of Aedes albopictus populations already resistant to DDT,
from Asia.
It is almost certain that
the success of Aedes albopictus in its 'world tour' was
due to passive transfer in the last 30-40 years, resulting the spread
of this insect to all the continents, except Antarctica. In Africa (Cape Town), the first
time of finding living larvae was in 1989, in used tyres shipped from
Japan. Though the shipment was
disinfected, still, two years later the insect was also
detected in Nigeria, Cameroon,
Equatorial-Guinea and Gabon. The presence of Aedes albopictus
was indicated In Central and Latin America from the 1980s-1990s; the
established populations of Mexico and Brazil began to
colonize other countries too, in a southern and eastern direction, by
expansion. By the
end of the 18th century, the insect was identified in area Hawaii; the
continental US appearance was registered in 1985, in
Texas. From that time on, the presence of the insect was
indicated in sequence, in 866 counties of 26
states. In Europe, it
was Albania (1979) where the first signal of this
insect was given. Next stations in spread were the
Mediterraneum, and the countries of the near East. From here, the populations of Aedes albopictus reached regions of the Netherlands,
Switzerland, France, Belgium and Germany too, in northern direction and
delayed expansion.
It seems that the
winner of the competition between Aedes albopictus and Aedes aegypti is
the former, with
the ability of the eggs to keep diapause
(dormancy) in development as a response
to environmental situation less advantageous
(in temperature, in humidity, in precipitation...).
If environmental
situation turns back to normal, the eggs go on in their developmental
program. At gene level, the dormancy period is supported by the
transcription
profiles of two genes (Ae. albopictus Pepck, PCNA); are
they promising targets for vector control at gene
level?
[Bonizzoni M.
et al.(2013): The
invasive mosquito species Aedes
albopictus: current knowledge and future perspectives Trends
Parasitol. 29: 460–468. doi: 10.1016/j.pt.2013.07.003]

The
drawbacks of using chemical insecticides (issues of
> selectivity often missing in targeting >
environmental
damage caused
> incorporation into food chain > resistance developed)
gave
boost to look for new ways in vector control, to restrict and
limit the growth of insect populations carrying infective
viruses.
4. Take a look at
some options provided by research in
physics, biology, and mathematics.
- SIT (sterile insect
technique):
sterility of insect males induced by ionizing radiation in
laboratory circumstances [mutations
provoked in reproductive cells too, at chromosomal
and gene
levels]; an open field release and a later recapture of these sterile
male populations make possible to follow their behaviour
in natural habitats.
Advantages
of the method: environmentally friendly, selective, no
opportunity for the next generation. Disadvantages of the method:
due to technical difficulties in application
> density dependent reactions in insect populations
> uneven
absorbed dose > physical status of irradiated males
restrict
their capabilities to compete with counterpart wild males in preventing
wild mating.

- RIDL
(release of insects carrying a dominant lethal),
a biotechnological version of SIT. A lethal transgene construct
with a dominant (developmental) trait repressed in laboratory
circumstances, integrated into insects'
genome.
Open field release of these insects results derepression of the
repressed
dominant trait, that is given to progeny generations
when mating in natural habitats. Thus, the derepressed
dominant
trait serves for preventing the offspring to reach its mature
adult form unwished.
- The
AeAct-4 (actin)
gene of the mosquito Aedes aegypti is
responsible for the regulation of wing muscles movements; the
expression of the gene is enhanced in females (the so-called "buzzy
oscillation" attracting males). After integration into larval
genome of a transgene contruct with AeAct-4 (actin)
gene promoter + intron sequences in it, the
developing females showed failure in
wing movements.
This is a lethal trait, since females with no wing movements are not
capable to escape from danger or to find food any more
[Guoliang Fu
et al.(2010): Female-specific
flightless phenotype for mosquito control
Proc.Natl.Acad.Sci.USA. 107: 4550-4554.].
- The method above was
elaborated for mosquito Aedes albopictus, too.
After integration into larval genome of a dominant lethal transgene
construct with AeAlbAct-4
gene promoter sequences in it, the developing females were
characterized by failure in wing movements. It was concluded,
that promoter sequences of Act-4
(actin) genes regulating wing movements in the two mosquito species,
are interchangeable [Labbé
G.M.C.
et al.(2012): Female-Specific Flightless
(fsRIDL) Phenotype for Control of Aedes albopictus
PLoS Negl
Trop Dis 6(7): e1724. doi:10.1371/journal.pntd.0001724].
- Open
field dispersal study on the competitive fitness in mating of
genetically modified insect males: the
OX513A-My1 sterile male population of Aedes aegypti carrying
a dominant lethal transgene construct with fluorescent marker included,
was released with wild counterparts in a
forest area with no inhabitants in Malaysia, on december 21, 2010
daytime, at temperature 32,6 oC,
relative humidity 66%, air movement 1m/s,
without precipitation.
Recapture was made in the period of december 22, 2010 to
january
5, 2011,
at a rate of 50% and 17% of mutant and wild
populations,
respectively.
Results: the estimated maximum distance after release was 250
m; the average distance in mutant populations
was slightly
less // the estimated average lifespan was 2 days
(mutant) and 2,2 days (wild), meaning, that due to transgene construct,
no significant alteration of this parameter was observed. Repeats of
dispersal experiments are planned in urban environment with female
mosquitoes' habitats. Main purpose is the reduction of vector
populations carrying infective agents > dominant lethal
alleles
are placed in the progeny by sterile males at mating, therefore, the
offspring does not reach its mature adult form.
- Dispersal
experiment similar to above, performed in suburb of Juazeiro,
Brazil, and published
in 2015
[Winskill P. et al.(2015): Dispersal
of Engineered Male Aedes aegypti Mosquitoes PLoS Negl Trop
Dis
9(11): e0004156. doi:10.1371/journal.pntd.0004156]. Using
multivariate analysis, the authors searched for optimum
description of mutant dispersal.

- Gene
silencing by
double stranded/dsRNA
sequences induced small interfering RNA/siRNAs.
As for the unpredictable evolutionary consequences of transgene
solutions, the regulatory insufficiencies in this field, the public
aversion towards it, they all, opened door to new biological
approaches in vector control.
In 2 days old Aedes
aegypti the
testis specific genes determining male character (10 genes) and the
gene determining female character were identified by subtractive
hybridization. Total RNAs of pupal and mature developmental
forms
were isolated for obtaining dsRNA sequences targeting the
previously identified specific genes determining the male and female
characters.
Larvae (with bacterial vehicle) and pupae (by injection) were fed with
the target sequence specific dsRNAs obtained. The
dsRNAs induced siRNA functions (transcriptional/post-transcriptional
shutdown) on target gene sequences ended up in the
stable
manifestation of sterile male and inhibited female (no sex)
character. Advantages
of the method:
no need for irradiation, for transgene constructs, for selection of
males or females. The method is based on molecular protective
classic mechanisms of eukaryotes, mechanisms specific for target
sequences so as the vital functions of the resulting insects with no
sex determination are retained, expectedly leading to
competitive
fitness in mating. The method is transferable to large scale production
and for other species in vector control. [Whyard S.
et al. (2015): Silencing the buzz: a new approach
to
population suppression of mosquitoes by feeding larvae double-stranded
RNAs Parasit Vectors. 2015; 8: 96. doi:
10.1186/s13071-015-0716-6.]
- RNA
guided CRISPR/Cas9
mediated genome
editing (type II): an adaptive protecting
mechanism in prokaryotes leading to cleavage,
heritable editing of
foreign nucleotide sequences (plasmid, virus) integrated in the
prokaryote genome. Participants of the mechanism are the
following: CRISPR (Clustered
Regularly
Interspaced
Short Palindromic Repeats) and Cas9
(CRISPR
associated 9
≡ endonuclease cleaving dsDNA). Principle of the
method: transcription of CRISPR interspaced sequences and
adjacent
short palindromic sequences into total RNA > formation of
transactivating tracrRNA from total RNA
> with
the
help of tracrRNA
formation of
crRNA
complementary to
interspace sequences
from
total RNA >
hybridization of tracrRNA to complementary palindromic
sequences
> activation of RNase III enzyme > tracrRNA
and crRNA
liberated, form association complex with enzyme Cas9 >
complementary
to crRNA interspaced dsDNA is the target for enzymatic
cleavage by
complex
Cas9
>
dsDNA breaks are repaired
by DNA repair machinery using nonhomologous end joining.
Result:
deletion, insertion (> mutation) and a disrupted target
gene. Advantages of the
method:
the result of cleavages on integrated plasmid /virus sequences
is inherited to offspring ('genetic memory'). According to experimental
results so far, the plasmid construct
CRISPR/Cas9 prepared for Aedes
aegypti needs
further development. [Dong S. et al.(2015): Heritable
CRISPR/Cas9-Mediated Genome Editing in the Yellow Fever Mosquito, Aedes
aegypti
PLoS
One. 2015; 10(3): e0122353.doi: 10.1371/journal.pone.0122353]

- Biocontrol
with cytoplasmic endosymbiont
bacteria Wolbachia pipientis. The supporting evidence as
below.
1.
Arbovirus (arthropode borne virus) spread depends on
the interactions among the virus, the insect vector and
several microbial components. According
to observations, Drosophila C (RNA) virus infection fatal to Wolbachia-free
populations of
Drosophila melanogaster
(fruit fly), results in delay or decrease of insect deaths, and also
of virus accumulation in fruit flies with Wolbachia
endosymbionts.
Restricted
to RNA viruses, the Wolbachia protection against viruses is based on
its high cytoplasmic density both in somatic and reproductive cells. A
competition among the parasite virus, the cytoplasmic endosymbiont
bacterium and the vector/host cell (of Drosophila, of
mosquito),
in reaching nutrition and energy source is presumed to manifest, giving
evolutionary advantage to Wolbachia of high density.
The mechanism of Wolbachia protection against (RNA)
viruses is related to generation of reactive oxygen intermediates
(ROI), to enhanced activity of regulatory miRNAs (in gene silencing),
and to the immune response provoked in the vector organism. [Johnson
K.N. (2015): The
Impact of
Wolbachia on Virus Infection
in Mosquitoes
Viruses 7:
5705–5717. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4664976/]
- 2.
It was in 1924 when Wolbachia was first demonstrated in mosquitoes
(Culex
pipens); however, for different microbiomes in different mosquito
genera, Wolbachia could not inhabit all of them. The introduction of
Wolbachia in biocontrol of insect vectors carrying pathogenic viruses
is based on the endosymbiontic capability to manipulate the
reproduction of the host organism by prioritizing its own bacterial
reproduction. This leads to deformities induced in the host:
to
parthenogenesis,
to feminization (> cytoplasmic thrive in vertical
transmission), to killing of males (> failures in DNA packaging
of
male reproductive cells), to cytoplasmic incompatibility
(offspring free of Wolbachia goes through deleterious embryonic
development).
- Among others, mosquitoes Aedes aegypti
and Aedes albopictus were
'transinfected' transiently and stably as well, with Wolbachia
strains of diverse activity, of Drosophila origin. Compared
to wild populations, the behaviour pattern and the "fitness"
(competitive mating) of the 'transinfected' insect populations were
studied.
The conclusions
denote the differences in results obtained from 'transinfections' with
different strains of Wolbachia. [Jeffries C.L., Walker T.
(2016): Wolbachia
Biocontrol Strategies for Arboviral Diseases and the Potential
Influence of Resident Wolbachia Strains in Mosquitoes Curr Trop Med
Rep. 2016; 3: 20–25. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4757633/]
- Emerging
questions:
- 1.
At an evolutionary scale, what kind of maneuvres will be deployed by
members of the triad: the pathogenic virus - the endosymbiont Wolbachia
- the insect vector, to
surpass 'transinfection' pressure?
- 2.
Spread of the
endosymbiont Wolbachia is realized in horizontal (somatic) and
vertical (reproductive) transfer, too.
Regarding 'transinfected' hosts, what are the cargo
and, how and where are they taken by Wolbachia in spread?



June,
2016
A report was published on the preventive effect of
interferon induced transmembrane proteins (IFITM) in early
phase
of Zika infection in cultures of HeLa, Vero, and A549
cells stably transduced with a lentivirus-IFITM construct [Savidis
G. et al.(2016/): The IFITMs Inhibit Zika Virus Replication
DOI: http://dx.doi.org/10.1016/j.celrep.2016.05.074]. According to the report, it was IFITM1 and IFITM3 of the IFITM proteins restricting pathogenic viral
infections
that showed effects as expected. Based on the cellular level
of virus-RNA measured, IFITM3 proved to be stronger
in
preventing early phase of virus infection.
As for the mechanism of prevention, authors of the report suggest a
direct action of IFITM3 on cell membrane and/or virus membrane,
interfering thus with membrane fusion and pore
formation so that viral genome and accessory key enzymes
cannot enter the host cell. Regarding that gene expression of IFITM
proteins at basic level is the highest in case of IFITM3, a protection performed by
IFITM3 in the early phase of Zika infection is presumed. Further,
IFITM3 may also act as
a spark in starting interferon induced chain reaction of gene
expressions. Considered as components of the innate immunity, IFITM proteins are able to
modulate significantly the early and decisive phase of pathogenic virus
infection. Confirming studies are
still needed (e.g. establishment of IFITM-deficient in vivo models).

June-July-August, 2016
Recent preclinical results in Zika vaccine development point at the efficacy of experimental plasmid DNA vaccine
{Zika
virus
immunogen coding expression
system (complete: virus pre-membrane + Env components, partial: only-Env component)}, and at the efficacy of inactivated Zika
virus vaccine {conventional
vaccine} in tests performed in mice and in Rhesus
monkeys models.
Zika source for the animal tests were: virus
isolates of the Asian
ZIKV16 line (ZIKV-BR/Brazil
ZKV2015 > BeH815744 and
ZIKV-PR/Puerto Rico > PRVABC59), differing in 5
aminoacids .
Mouse Model
- → Immunogenic
studies: Balb/c
mice (5-10/group) immunized once (50
μg DNA vaccine/i.m.). For detection of specific antibodies
ELISA tests were performed on week 3 after immunization.
- Observations:
- Compared
to the only-Env-DNA
vaccine, the complete (pre-membrane + Env) DNA vaccine
provoked higher
titers in Env specific antibodies.
No antibody response specific to the pre-membrane
component (alone) was demonstrated.
- Zika
virus specific
neutralizing antibodies were generated by the complete (pre-membrane + Env) DNA vaccine (microneutralizing
test).
- Env-specific TCD4+
and TCD8+ lymphocyte
response was induced by the complete (pre-membrane + Env) DNA
vaccine (IFN-γ
ELISPOT test + multiparametric intracellular cytokine
staining).
- → Evidence
for the protective effect of DNA
vaccine: virus challenge to
Balb/c mice after immunization (week 4) and to control mice
{i.v. ZIKV-BR or ZIKV-PR / 105
virus (102 PFU)}.
Quantity of viral load was
controlled by RT-PCR.
- Observations:
- In control animals
viremia was established in 6 days.
- ZIKV-BR
viremia was prevented (virus
number < 100 copies/ml) at
different test times (n
=
10) by complete (pre-membrane + Env) DNA vaccine.
Similarly, complete protection was observed following virus challenge
on week 8 after immunization. Protection
was also conferred by complete
(pre-membrane + Env) DNA vaccine when
virus challenge with ZIKV-PR strain was performed.
- → Immunogenic
studies:
Balb/c mice (5/group) immunized
once
with inactivated Zika virus
vaccine
(1
μg / i.m. or s.c., strain
Puerto Rico PRVABC59). For detection of generated antibodies,
ELISA tests were performed.
- Observations:
- Compared to inactivated Zika virus
vaccine/s.c., the
inactivated Zika
virus vaccine/i.m. resulted higher antibody
titers
in the immunized animals. Both types of delivery succeeded in
generating Zika
virus specific neutralizing antibodies.
- Parenteral
(i.v.) virus challenge (ZIKV-BR strain) on week 4 after immunization;
complete protection was achieved in animals immunized by the intramuscular route.
- →
Adoptive transfer: IVIG (intravenous immunoglobulin)
- Given to recipients, IgG
purified from
sera of Balb/c mice immunized with complete
(pre-membrane + Env) DNA vaccine
(ZIKV-BR)
provided antiviral
protection at a strength proportional to the Env specific antibody
titer of the immunized donor.
In other words: antiviral protection is conferred by IgG on
condition the Env specific
antibody titer exceeds a critical threshold level.
[Larocca R.A.,
Abbink P. (+24) (2016): Vaccine protection against Zika virus from
Brazil
Nature Letter Accelerated Article Preview
published online 28 June 2016]
Rhesus
Model
- → Immunogenic
studies: inactivated Zika virus
vaccine (s.c.) to
16 Rhesus monkeys (5 μg/animal) on
week 0 and 4. The generated antibodies were checked
by ELISA method.
- Two weeks after the first
immunization the
rise of Env specific
antibodies and Zika
virus specific
neutralizing antibodies (mikroneutralizációs teszt) were
detected
in the animals. Following
the second immunization (week 4), on week 6, the antibody titer got
even higher. Compared to control animals, in majority of the
immunized monkeys, Env
specific moderate cellular immune response was elicited (IFN-γ ELISPOT
test).
- → Evidence
for the protective effect of inactivated Zika virus vaccine:
virus challenge {ZIKV-BR
or ZIKV-PR / 106
virus (103 PFU)}
to immunized and control
Rhesus monkeys (s.c.).
Quantity of viral load in host animals was detected
by RT-PCR, virus infectivity was confirmed in Vero
cell
culture.
- Observations:
- In
control monkeys viremia was established within 6-7 days after
virus challenge. Virus was detected in urine, in cerebrospinal fluid
(CSF) on day 3; in colorectal and cervicovaginal secretions on
day
7. No significant
difference was shown in plasma viral load following ZIKV-BR
or ZIKV-PR challenge.
- Complete protection of monkeys
against Zika infection was provided by inactivated Zika virus vaccine (virus
number in blood /urine /CSF/colorectal secretions /cervicovaginal
secretions
< 100 copies/ml).
- → Adoptive
transfer: IVIG
- Plasma IgG
was purified (protein G affinity chromatography) from monkeys immunized with inactivated Zika virus vaccine (week 8 after
immunization). Balb/c
mice
were infused with the
IgG obtained. Virus
challenge {ZIKV-BR
/105
virus (102 PFU)}
to mice following infusion: proportional to IgG dose
infused, protection against Zika (high dose) and decreased
viremia
(low dose) were observed.
- Naïve
Rhesus monkeys were
infused with IgG as above. Following virus challenge
{ZIKV-BR/106
virus (103 PFU)}, one of the
recipients given high dose
IgG showed complete protection against Zika, while the other of the
recipients showed slight viremia on days
3-5. Virus replication was not supported by IgG in subtherapeutic
doses.
- In
brief: according to teaching of experiments in rodents and primates,
adoptive transfer of IgG from donors immunized with inactivated Zika virus vaccine
is promising tool for conferring protection against Zika.
[Abbink
P., Larocca R.A. (+ 36) (2016): Protective efficacy of multiple
vaccine platforms against Zika virus challenge in rhesus monkeys
Science 04 Aug 2016:]


September,
2016
WHO Situation Report comprising data up to
14th September, notifies about the so far revealed two lineages of Zika
virus, the lineage
Zika-Africa and the
lineage Zika-Asia.
- Lineage
Africa is observed in Africa; seven new registered
infections were characterized by nucleic acid sequencing, in Guinea-Bissau.
- Strains
of lineage Asia are
documented in Asia, in Western Pacific Region, and in the Americas.
- Neurological
complications are related to after-2007-strains of Zika-
Asia. However, considerations should be extended to
any
strains/lineages, due to shortages in current diagnostics and
knowledge.
- After-2007-strains of
Zika-Asia were isolated in French Polynesia (since 2013), in the
Americas (since 2015), in Cabo Verde (2016).
- Since 2007 altogether 72
countries/territories have announced mosquito mediated Zika infection.
70 countries/territories
of them gave the first infection signal in 2015.
- Since
February, 2016 we are aware of Zika infection transmitted by
non-mosquito (human) vector (signalled in five countries of the
Americas, in six countries of the European continent, and, in a country
of the Western Pacific Region).
- The
potential relationship between congenital malformation of the central
nervous system and Zika virus infection is underpinned by reports
from 20 countries/territories.
According
to Zika molecular evolution,
the process
has progressed by
genetic recombinations resulting
three distinct genotypes so far; the East-African
cluster (cca. period 1892-1943), the West-African
cluster (evolved in several streams of cca. 1935-1940; cca. 1950-1960;
cca. 1965-1985, cca. 1995),
and the Asian cluster (cca. 1960). As for phylogenetic
analyses,
the spread of virus from East Africa to direction West Africa and Asia,
then to Western Pacific Region and the Americas, is presumed.
Early in 2015 consideration:
genome of Zika virus settled in North-Brazil is comparable
with the genome of Zika virus isolated in French Polynesia, in
2013. This latter belongs to the Asian cluster.
December-2015 consideration: nucleic acid sequencing
(NGS = next generation
sequencing) of Zika
virus settled in Guatemala
and Puerto Rico, further, their evolutionary estimation
(GenBank KU501215/Puerto
Rico PRV-ABC59; GenBank KU501216/Guatemala 8375;
GenBank KU501217/Guatemala 103344) support the notion that
Zika genomes of Guatemala and Puerto Rico strains are similar to Zika
genomes of North-Brazil
and French Polynesia strains; they all belong to the Asian cluster.
Nucleic acid similarity
among Zika virus genomes isolated in the western
hemisphere, are >99%.
Nucleic
acid similarity among Zika virus genomes isolated in the
western
hemisphere, in East Africa, in West Africa, show about
89% similarity.

October-November,
2016
WHO Situation Report, 13
October 2016 (for comparison see WHO
Situation Report, 15 September 2016)
- Since 2007 altogether 73
countries/territories have announced mosquito-mediated Zika
infection.
→ In September the number was 72
countries/terrritories.
- 67 countries/territories of the 73
mentioned gave the first signal of infection in 2015.
→ In September the number was 70
countries/territories.
- The
potential relationship between congenital malformation of the central
nervous system and Zika virus infection is suggested by reports
from 22 countries/territories.
→ In September the number was 20
countries/territories.
Related to the latter, to
Zika neurotropism, the potential far reaching consequences of it
provoked debates unfolding significant diversities in professional standpoints. Two examples of them are shown below.
Summarizing thoughts/1
As for international publications registered in PubMed in time
period running from the first detection of Zika virus in humans in
Niger 1950, till 27 May
2016, the authors are
not convinced about the
causal relationship presumed between Zika virus infection
and multifactorial congenital microcephaly.
Questions
raised, links missed
?
thorough tracking of pregnancies exposed to Zika virus;
detailed
laboratory tests for detection of virus and of
immune analytes; use of
ultrasound
examinations ?
?
economic environment and conditions of public nutrition in
countries involved in virus epidemic ?
?
interference of Zika with other virus-, bacterial, parasite
infections ?
? standards in microcephaly diagnostics ?
?
virus mutations resulting the accelerated epidemic
of autumn 2015, expanded later throughout
the continent of Latin America ?
? mechanisms explaining Zika neurotropism ?
Summarizing thoughts/2
Opposite to Summarizing thoughts/1 while evaluating international studies registered in PubMed,
the authors here are less reserved in adopting the causal relationship
presumed between Zika virus infection and multifactorial
congenital
microcephaly. The preliminary
report on the ongoing studies in Brazil is taken as supporting
rationale.
As
for the authors, the causal relationship is evidenced
by detecting
virus particles in placental chorionic villi and in brain tissues.
However, Jing Wu et al. pointed on the lack of
laboratory tests periodically performed on blood
and urine
samples.
Similarly, on basis of studies published so far, the pathogenesis
of Zika microcephaly is classified unresolved as for Jing Wu et al
(Summary/1).
Dissenting from the opinion of Jin-Na Wang et
Feng Ling, Jing Wu et al. consider the experiments on mice not
comparable to human cases.
Both summarizing thoughts
urge the involvement of non-human primates in further experiments.
Studies
were run in eight public hospitals (Recife, Brazília) on 32 newborns
with microcephaly and
62 healthy newborns (controls chosen by the parameters of: same living
areas, expectedly same date of birth, without microcephaly). Among the
32 newborns with microcephaly, in 13 (41%) babies the Zika infection
was confirmed by laboratory tests. No Zika infection was detected in
control babies.
Analyses
of sera and cerebrospinal fluid samples for detecting Zika
specific IgM and detecting Zika virus by serology and
quantitative RT-PCR, respectively.
Analyses of maternal sera by Zika and Dengue virus
specific plaque reduction neutralizing test (PRNT).
In progress: prospective
cohort studies in support of the presumed causal relationship
between Zika infection and developing congenital microcephaly.
Preliminary reports
on the ongoing studies are found here:
Brazil
[Brasil P, Pereira JP, Jr, Raja
Gabaglia C, Damasceno L, Wakimoto M, Ribeiro Nogueira RM, et
al.(+14): Zika virus infection in pregnant women in Rio de
Janeiro – Preliminary report. N
Engl J Med. 2016 [Epub ahead of print]. doi:
10.1056/NEJMoa1602412.]
Colombia
November 2016:
recruiting for Phase 1 clinical trial No.
NCT02963909
First
Zika vaccine candidate in human clinical trial: alum-adjuvated
Zika virus Purified Inactivated Vaccine
(ZPIV) in Phase 1 clinical trial.
Randomized,
double-blinded,
placebo-controlled trial for testing prevention, safety,
efficacy, immunogenicity in healthy
(Flavivirus-naïf and
Flavivirus-primed), aged 18-49 volunteers of both genders. Estimated
date of final data collection is May, 2018.
(Source: ClinicalTrials.gov)

November-December,
2016
Zika - Dengue cross-neutralizing
antibodies in
preventing Zika infection
X-ray crystallographic
and cryo-electronmicroscopic studies evidenced the dynamic changes in
the conformation of Zika
and Dengue surface
'E' (envelope)
glycoproteins
in early phase of virus infection. These dynamic changes manifest in the course
of specific interactions between virus
and target cell
surfaces (virus adhesion, virus docking), then in the course
of membrane fusion
carried about by virus surface and target cell (or
cell
compartment) membranes.
The membrane fusion is a step critical and decisive in the
process of virus infection. Regarding experimental data, membrane
fusion mentioned above is
inhibited by Zika-Dengue cross-neutralizing antibodies isolated
from patient with Dengue infection,
having "locking effect" on actual conformation of
virus 'E' glycoproteins ("super
serogroup") in surface topography.
The results so far may give support to simultaneous prevention of
Dengue and Zika infections (vaccine development?) as well
as to
a reliably broadened diagnostic
window (serodiagnostic test development?).
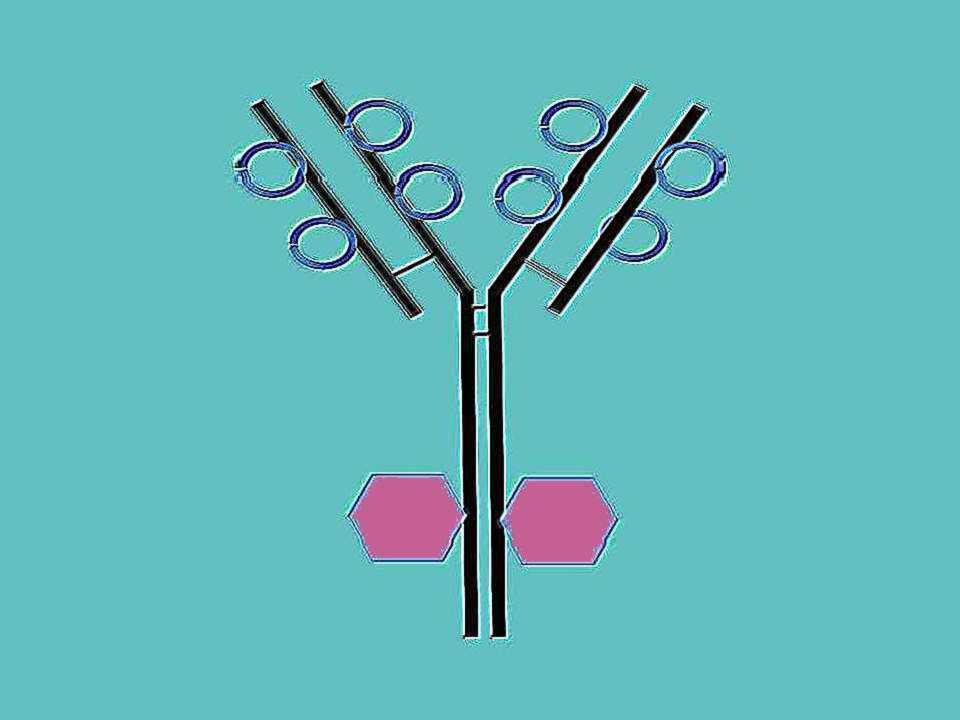
Evidence 1
Neutralizing all four Dengue serotypes,
broadly neutralizing
human antibody to Dengue virus (to envelope 'E' glycoprotein
dimer epitopes -EDE-) is
also efficient in neutralizing Zika infection in vitro, in Vero cell
cultures.
[Barba-Spaeth
G. et al. (+15): Structural basis of potent Zika–dengue virus antibody
cross-neutralization Nature 536, 48–53 (2016)
doi:10.1038/nature18938]
Evidence 2
H
and L chain variable region sequences of human antibodies to Dengue
virus cloned into plasmids > plasmids for transient transfection
of
human
HEK293T cells > purification of monoclonal
soluble antibody C10 produced > preparation of IgG-Fab (antigen binding) fragments
> in
vitro reaction of IgG-Fab
fragments with Zika virus
(pH=8.0; pH= 6.5;
pH=5.0) >>> cryo-electronmicroscopy of the
interaction
between 'E' glycoprotein dimer(s) + Fab(s).
[Zhang S. et al. (+10): Neutralization mechanism of a highly potent
antibody against Zika virus Nature Communications 7, Article
number: 13679 (2016)
doi:10.1038/ncomms13679]

 In
Focus: HIV/AIDS
virus
In
Focus: HIV/AIDS
virus
History
- The
HIV (human immunodeficiency virus) is an icosaeder
enveloped virus (family Retroviridae / genus Lentivirus / 9749
nucleotides - two copies of single (+)RNA
strands in the genome) with surface spikes composed of glycoprotein
(gp) trimers built up from gp120/gp41 dimers in special
spatial
arrangement. The binding of the virus to the target cell is mediated by
these surface glikoprotein spikes. During productive infection
it
is the viral surface gp120/gp41
glycoproteins that bind to surface receptors and coreceptors (CD4 …
chemokine receptor CCR5
… chemokine receptor CXCR4) on cellular
elements
of innate and adaptive immune responses (macrophages,
dendritic cells,
CD4+ T lymphocytes).
After fusion of the viral membrane with the target cell membrane along
with the activation of post-receptor signalization, the target cell is
further restrained in its vital functions, ultimately leading
to
cell damage. The process is amplified by the multiplication (reverse
transcription) of viruses
accompanied by mutations in infected
cells
and, virions liberated from living and dead cells continue to infect
other target cells, too.
Finally, in majority of cases, after latency time, the resulting virus
load ends up in fatal immunodeficiency of the host organism
(Aquired Immunodeficiency Syndrome - AIDS).
- An
alternative to
HIV is the SIV (simian
immunodeficiency virus) targeting primates. The SIV infection, mainly
in apes, leads to a condition similar to that of the HIV
infection
mentioned before, mostly in Africa. The virus strain SIVcpz
detected in chimpanzee
in 1999, was genetically almost identical to human HIV-1 virus and, the
SIV virus detected in Cercopithecus (Old World monkeys) was
genetically
closely related to HIV-2 virus.
The
virus
strain SIVcpz is suggested to evolve from the
recombination
of two different SIV strains
(after horizontal transfer). It is supposed that smaller
monkeys
infected with SIV strains different in their genomes were the prey
animals for predator chimpanzee. Since the smaller prey
animals
were eaten,
the predator chimp became a carrier
of the different virus
strains consumed with the preys. Consequently, the SIV strains carried
by the predator were free to combine with one another in their new host
and,
after a further increase in virulence, they
could cross
the chimp-human barrier as well, in a prey-predator manner
similar to that mentioned
before.
This last transmission may have happened around 1920, in Kinshasa - DR
Congo. After transmission to the human body, these SIV
strains continued
their propagation as HIV, in diverse lineages of HIV-1
virus (HIV-1 M,N,O,P) and, in subtypes (A,B,C,D,F,G,H,J,K)
of
the most frequent M lineage
of HIV-1.
- Even
if different from
HIV-1, the
HIV-2
virus may have arrived in the human body in a similar
"nutritive"
way, however, in this case the Cercopithecus is supposed to serve as
the prey animal. Since its first detection, the
geographical spread of
HIV-2 virus has been mainly localized in West Africa (Bissau-Guinea,
Senegal). In the majority of cases, HIV-2 infections do not end
in AIDS syndrome. If it happened at all, the overwhelming
plurality of HIV-1 virions could be detected in the patient.
- According
to
biogeographical
data, it is the M lineage ’B’ subtype
of HIV-1
virus to
be detected most often in our days.
-

According to
data in the WHO UNAIDS booklet of 2015, the number of
HIV
infections registered in 2014 was 36.9 million.
Among
them 17.1 million were not aware of their infected status.
Although a global decrease in the number of infections could
be
seen, still,
there were 2 million new infections and 1.2 million deaths of
AIDS reported in the subject year 2014.
[http://www.unaids.org/sites/default/files/media_asset/AIDS_by_the_numbers_2015_en.pdf]
Which
was unbeleivable 15 years ago: approximately 15.8
million of HIV
infected had access to antiretroviral therapy, according to the report from June
2015.
Calculating with the access to antiretroviral therapy, the main goal
declared by WHO to reach until 2030 is the global abolishment of the
AIDS epidemic. Meanwhile this main program, the interim goal until 2020
is that 90% of the infected be aware of their infected
status, 90% of the latter have access to
antiretroviral therapy, and in 90% of the latter the antiretroviral
therapy result a
significant decrease in virus number. A further interim goal
is
that the current number of new infections decrease by 75%
and, the
number of intrauterine and postnatal (breast feeding) infections
(vertical transmissions) reach and remain at zero level.
Calculating
with the infected regions,
and with emphasis on low income countries, the estimated cost
of reaching interim goals by 2020 is 31.1
billion US
$, a peak in costs expectedly reduced in later phases of the program.
The UNAIDS
data reporting a decrease in the number of HIV infections highlight
the availability of current antiretroviral therapy and
suggest further development of adequate
drugs targeting
virus-cell interactions or critical steps in virus replication. Even
so, the cardinal aim of our days in the fight against HIV/AIDS
is
the development of neutralizing antibodies against the HIV virus. These
antibodies -vaccines- supplementing the actual antiretroviral therapy,
could be a choice in the prenatal and postanatal care of
infections, and mostly, in the prophylaxis of infections. This latter
undoubtedly could generate a positive move in the economic
indicators of the countries concerned. The greatest challenge in
vaccine development is the highly varying character of the antigen
determinants in the HIV virus envelope (increased virus growth rate
accompanied by increased mutation rate resulting in great variety
of viral surface epitopes).
In
harmony with those above, interesting new approaches in
vaccine research and development were
published. [Summarized
in > So Youn Shin (2016): Recent
update in HIV vaccine development Clin.Exp.Vaccine Res.
http://dx.doi.org/10.7774/cevr.2016.5.1.6.]
A
remarkable approach is based on
the well-known observation
that in
20% of HIV infected patients, 2-3 years after infection, so
called
broadly neutralizing antibodies (=bNAbs) could be detected in the
blood. These antibodies due to the 2-3 years of evolutionary selection
(clonal selection) became capable to neutralize virus isolates
harboring different surface antigens. Introduced into primates via
passive transfer, the bNAbs restrained virus progression in infected
animals, and prevented new infection in healthy ones. No data are
available of the efficacy of bNAbs via passive tranfer, in
humans.
Taken
together, the generation of bNAbs by active immunization
became
the goal to achieve. For the experimental production of bNAbs, the
immunogen substance was assembled from different, artificially
constructed HIV envelope protein components. It turned out,
that the success in constructing and in immunizing with the
HIV
envelope protein components, depends on the binding of the first
immunogenic component to the germ line B cells, on the activation of
the rare precursor B cells potentially capable to develop
into B
cells producing bNAbs. In the publication the construction of
HIV
immunogen vaccine candidates are presented [Joseph G.
Jardine, Daniel W. Kulp, Colin Havenar-Daughton (+19) (2016):
HIV-1
broadly neutralizing antibody precursor B cells revealed by
germline-targeting
immunogen. Science: 351:
1458-1463].
Another
remarkable
approach was published two years earlier.
In its content a dynamic, other than usual way of thinking,
a paradigm shift is disclosed.
This
research was based on the observation that HIV
infection at body temperature (37 °C) is brought
about by the
interaction of virus and target cell surface glycoprotein epitopes (gp120/gp41 … CD4 … CCr5/CXCR4
ternary complex), characterized
by continuous and dynamic change in their spatial appearances (variations
in spatial conformation, induced exposure or hiding, approaching to...
or,
move away from...), before membrane fusion (prefusion). This continuous
and dynamic change in surface epitopes results in transient and
short-lived prefusion surface patterns hence, the immune
system
has no time enough for recognition and for generation of
specific antibodies in response (a possible cause of failure
in the development of previous vaccines).
Since the interaction and later the fusion, of virus surface
and target cell membrane are temperature dependent
processes,
the temperature guided (<25°C) synchronous arrest of the prefusion
surface pattern results in the fixation of
the transient interactions of surface epitopes,
preventing thus the fusion of membranes.
The considerations
above led to the
implementation of a coculture system, in which virus donor B cells were
cocultured with virus acceptor T cells, in vitro.
After the
prefusion synchronous arrest at different temperatures (<25°C), the surface
patterns of epitopes were fixed by 0,05% formaldehyde.
Mice were immunized with this "designed antigens". The monoclonal
antibodies produced by the animals did not react with the
"static", classic virus epitopes. According to expectations, the bNAbs
produced by these "designed antigens" help to avoid virus-target cell
membrane fusion, the step initiating productive HIV
infection.

This
method of obtaining "designed antigen" may be beneficial for
other
viruses, too. A further advantage is its applicability
in developing topical microbicides.
[Tuckweng Kok,
Adriana
Gaeguta, John Finnie, Paul R Gorry, Melissa Churchill and Peng Li
(2014): Designer
antigens for elicitation of broadly neutralizing antibodies against
HIV.
Clinical & Translational Immunology
3, e24; http://www.nature.com/cti/journal/v3/n9/full/cti201422a.html]
An alternative
to prefusion
synchronous arrest is the research published two years
later (in 2016),
describing the crosslinking (by glutaraldehyde, further, by EDC/NHS
= [1-ethyl-3-(3-dimethylaminopropyl)carbodiimide
hydrochloride/N-hydroxysuccinimide])
of antigenic trimers (> surface and transmembrane
glycoproteins) of virus surface envelope, to stabilize their
conformational flexibility in a
native-like form. The
latter makes it possible for neutralizing antibodies
(bNAbs) to access and,
bind to corresponding antigens of the crosslinked
complex. This binding is
confirmed, and the heterogenous population of antibodies is separated
into fractions, by affinity chromatography, wherein weak or non-neutralizing
antibodies are not bound to antigenic complexes stabilized
by crosslinking agents. The publication gives
support to the idea that aiming at vaccine development,
the stabilization of flexible surface antigenic
determinants is inevitable for obtaining neutralizing antibodies
(bNAbs) full of function. [Schiffner
T. et al (2016): Chemical Cross-Linking Stabilizes Native-Like HIV-1
Envelope Glycoprotein Trimer Antigens J.Virol. 90: 813-828.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4702668/]
As
long as
there is no effective HIV vaccine...
- Clinical efficacy of vaccine
candidates: collected data (2013) ...
- IFE-UNAIDS
economic and ergonomic considerations for low and middle income
countries from point of HIV vaccination ...
- January, 2015 - January, 2017, South Africa, 6 clinical
centers: HVTN100
Phase
I/II randomized, double blind, placebo controlled, parallel
clinical trial, enrolling age range 18-40 of both genders, with primary
purpose prevention
...
The
latest WHO and UNAIDS guidelines [WHO
Guideline on when to start antiretroviral therapy and on
pre-exposure prophylaxis for HIV. World Health Organization, Geneva;
2015] [UNAIDS
Strategy for 2016—2021: fast-tracking to zero. UNAIDS,
Geneva; 2015]
are exemplified in a Danish study comprising 20 years
follow-up of serodiscordant pairs (man + man in pair, one of
them
infected) in HIV infected communities. While the pairs were under
continuous laboratory check up, the infected persons of them were
given antiretroviral
drug treatment
consecutively, with no skips. Results of the long lasting
study were summarized and interpreted by mathematical and
epidemiological approaches as well [Okano
J.T. et
al. (2016):Testing the hypothesis that treatment can eliminate HIV: a
nationwide, population-based study of the Danish HIV epidemic in men
who have sex with men. Lancet Infect. Dis. 16:
789-796.]
It
was found that the threshold value to
eliminate HIV according to WHO and UNAIDS guidelines {≡ one
new HIV infection/1000 person/year},
can be approached by the combined
effect of the almost full coverage (92%)
in treatment of
infected communities and,
the consecutive adherence to antiretroviral therapy (∑ rate
of virus suppression = 98%).
The goals were reached by 'treatment as prevention' of HIV
infected persons to
decrease the risk of HIV transmission to their non-infected partners.
Population coverage was further supported by the changes along the 20
years in Danish public health practice. In the period
of 1996-2008
the eligible patients for antiretroviral therapy were those
with CD4+ lymphocyte number below the threshold of 300/µl.
This threshold was reset to
level 350/µl in 2008. From
2011 on, irrespective of
the CD4+ cell number, all
HIV infected persons
became eligible for antiretroviral drug therapy.
Results above highlight the alignment of
three optimal contributors to HIV elimination:
- at individuum level the compliance
with, and adherence to treatment,
- at community level the great coverage
(all infected involved in treatment, if possible),
- supporting biological
option is the use of drugs with retarded or no developing
resistance to
them.
Copyright©
www.bioteomed.org

Contact:
btm(at)bioteomed(dot)org
nature,
science, research, invention, industry, biology, physics,
chemistry, agriculture, medicine, drugs, patents, technology, literature, arts, education



 In
Focus: Zika
virus
In
Focus: Zika
virus
















 In
Focus: HIV/AIDS
virus
In
Focus: HIV/AIDS
virus





